Peculiar Paneer: Cheese Making! (Autumn, Elly, Kateri, Symone)
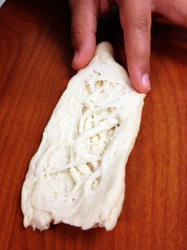
In our first Kitchen Chemistry experiment, our group was assigned to make Paneer in order to study the affects of protein denaturation. Some of you might be wondering what Paneer actually is, and basically it is an Indian cheese that is very simple to make. First, we weighed a quart of milk and poured it into a pot to let it warm up to 80°C (176°F). After it reached this temperature, we added 5 tablespoons of lemon juice and let it sit for half an hour. When we checked on the solution after the allotted time, we were surprised to find that the milk had become chunky. This was due to the adding of acidic lemon juice and heat, causing the proteins to denature and the milk to curdle! Next we strained the solution with a towel to catch all the curdled pieces, and squished and formed into a square to cut up and be served. We weighed our final solution and discovered that much of the mass had been lost in the process! This was because we had to discard the liquid that did not curdle, otherwise known as whey. Next we tasted it; the flavor and texture were definitely unique, and it had a slight lemon tang to it. Overall, our group couldn’t believe it was that easy to make cheese! So if you’re ever craving something cheesey and you want it to be easy, try making Paneer!
Twisted Pretzels (Allie, Katherine, Nicole, Trisha)
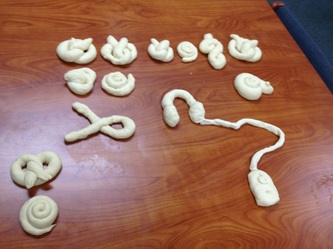
In this, our first installment to Kitchen Chemistry, Team Flour Power took on the challenge of pretzels and the browning affect that occurs when baking. Our amazing professor DBH sent us on the pursuit of finding the Golden Pretzel. Like any true sensei DBH set us up with all the tools of success, including the pretzel dough, all we had to do was shape our pretzels, give them a quick bath, bake, taste, and compare. While shaping the dough we may have gotten a little creative with the shapes, as you will see. After all dough was rolled and shaped we then dipped the pretzels into one of three baths; lemon juice and water for an acidic solution, water for a neutral solution, and a baking soda and water for a basic solution. We then sprinkled on a little salt and placed our creations into a 350-degree oven 8 minutes and awaited our results. After looking and tasting each pretzel we found that lemon juice kept the pretzels white but gave a nice crust with a rather doughy center. The water bath pretzels came out white as well and had a rather soft outside, and were more bread like. In the end the baking soda/water solution gave us our "Golden Pretzel" because the baking soda is basic it gave us our browning affect we were in search of, a nice crisp outside crust with a classic pretzel taste! So next time you're craving some pretzels make sure to dip or coat them with a baking soda/water solution to create the browning effect and get that golden pretzel.
Caramels and Paneer and a few dipNdots- Riane, Allie, Juliana, & Natalie
For our first Kitchen Chemistry assignment our group was designated to make both caramel and paneer. Paneer is a form of easy home made cheese. While making it, we learned about protein denaturation. Fist, we began by boiling .86 grams of milk. Once at boiling point (80 degrees celsius), we proceed by adding 3 tablespoons of citric acid. As you can see from the picture below, the milk slowly started to curdle and thicken. We allowed the curds to cool for a half an hour and then strained the cheese with a cloth. After straining it to get all the excess liquid out, we shaped it into a small rectangular shape. As an end result, we measured .045 grams of paneer. Due to the fact that we did want our paneer to taste too lemony, we did not add enough citric acid so it turned out a little more watery than would have been desired.
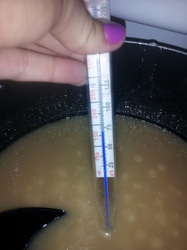
As for the caramels, it wasn't as difficult to make than expected. We started by mixing white sugar, brown sugar, corn syrup, evaporated milk, heavy whipping cream and butter in a pot and heated it up. The temperature has to get up to 250 degrees Fahrenheit and as the temperature rose the mixture of ingredients got more sticky and thicker.. Then once the temperature reached just under 200 degrees Fahrenheit, the caramels mixture started to bubble and all the air was escaping. After, it reached 250 degrees, we mixed in vanilla, put it in a pan and then froze it until it was cooled enough to cut in pieces. In the pictures you can see the temp rising and more bubbles popping up. The chemical process that happened with the caramels is called the Maillard reaction and caramelization. The caramels turned out good and you could taste the "browning" in them and they more chewy then expected.
As for the caramels, it wasn't as difficult to make than expected. We started by mixing white sugar, brown sugar, corn syrup, evaporated milk, heavy whipping cream and butter in a pot and heated it up. The temperature has to get up to 250 degrees Fahrenheit and as the temperature rose the mixture of ingredients got more sticky and thicker.. Then once the temperature reached just under 200 degrees Fahrenheit, the caramels mixture started to bubble and all the air was escaping. After, it reached 250 degrees, we mixed in vanilla, put it in a pan and then froze it until it was cooled enough to cut in pieces. In the pictures you can see the temp rising and more bubbles popping up. The chemical process that happened with the caramels is called the Maillard reaction and caramelization. The caramels turned out good and you could taste the "browning" in them and they more chewy then expected.
Bottomless Pit (Angie, Seth, Isaac, and Kristin) -Caramel Making.
Bottomless Pit Continued...
This is an image of the ingredients we added before we began the actual cooking part. In the pot is brown sugar, white sugar, butter, corn syrup, heavy cream, and whole milk. We put the burner on medium heat; we waited until the thermometer reached the temperature of 250 degrees to remove the pot from heat. The temperature began about 150 degrees, didn’t see much change until 224 degrees. The mixture started to become a rather white swirl mixture. Temperature then reached 213 degrees and the chemical process has started to slightly boil. Not very much but noticeable. Temperature is 224 degrees, the chemical process has reached its browning stage. The color has changed to a darker yellow color. This image shows a temperature of 232 degrees. The mixture has not changed at all since the past temperature of 224 degrees. At the temperature of 248 degrees the mixture becomes much more thick and the boiling is almost settled down and the mixture that is formed is dense. Immediately after we took the pan off of the heat we added a LOT of vanilla extract! We then poured the caramel mixture into a pan and put it into the fridge to dry. The finished product was amazing!
Dippin' Dots and Pretzels man!(Alex, Chad, Alec, Xai)
The day we did the kitchchem activity our group was responsible for making pretzels and experimenting with the browning of the pretzels. The first thing we had to do was to make shapes with the dough. Secondly, we made the shapes to dip it in two different solutions. One was water and baking soda and the other was water and lemon juice. We had to see the difference from the two solutions after the baking process was completed. We found out that dipping the dough in the baking soda and water solution, which is what most pretzel makers use, browns the dough while the lemon juice and water solution did not brown the pretzels but still gave them a crust and they still tasted like pretzels.